Biomonitoring challenges
The physico-chemical methods used to monitor the quality of the air, water or ground measure the concentrations of components present in the environment for comparison with regulatory values. However, these techniques fail to provide specific information about the effects of pollutants on organisms. Metrology systems will doubtlessly continue to improve, but biomonitoring (based on fungal or plant models) will always remain the only valid approach to assess the biological effects of an environmental alteration (Van Haluwyn et al., 2011).
The idea of observing the effects of air pollution on plant compartments dates back several centuries to J. Evelyn’s first description of leaf damage in 1661 following the contamination of air by coal combustion (Treshow & Bell, 2002). However, the real concept of plant and fungal biomonitoring of air quality dates back to the 19th century, when Nylander proposed using lichens as “hygiometers” of air quality, after noting that lichen colonies had disappeared in locations close to cities such as Paris (Nylander, 1866). Since then, biomonitoring has been used for various objectives, including the analysis of the spatial and temporal distributions of pollutant impacts or the identification and monitoring of isolated sources of contamination. This approach is also an increasingly common tool in health and ecological risk assessment methodologies and plays a central role in informing the general public and facilitating decision making in public policies (Cuny, 2012).
Biomonitoring and its various concepts were formalized by Garrec & Van Haluwyn (2002).
“Biomonitoring is the use of responses at all levels of biological organization (molecular, biochemical, cellular, physiological, tissue, morphological and ecological) of an organism or a set of organisms to predict and/or reveal an alteration of the environment and to follow its evolution”.
These approaches involve observing and globally measuring the response of organisms to contaminants. They take into account not only the combined effect of multiple compounds present in the environment, but also consider the ecological and climatic characteristics of the territory (Van Haluwyn, 1998).
A recent standardization process led to a European consensus on the definition of biomonitoring:
“Biomonitoring is the use of biological systems (organisms and communities of organisms) to track changes in the environment in space and/or over time” (Leblond et al., 2014).
This approach is not therefore a substitute for physico-chemical monitoring techniques, but is rather a supplementary source of data (Cuny et al., 2008). It has a number of advantages, since it uses multiple models that can show effects at different levels of biological organization. The most commonly used plant and fungal models are tobacco, bryophytes and lichens (for air pollutants), ryegrass, cabbage, clover and fungi (for soils), and elodea, lemnoideae and bryophytes for aquatic environments.
The complementarity of physico-chemical monitoring and biomonitoring of the air (Photo courtesy of APPA).
Biomonitoring approaches and concepts
Biomonitoring is based on two approaches.
In situ biomonitoring, also called the integrated or passive approach: this approach is based on the observation of organisms that are naturally present in the environment studied. It is mainly used to observe long-term impacts, and takes ecological and climatic conditions into account.
In active biomonitoring, organisms that have been cultivated under controlled conditions (e.g. a greenhouse) or were taken from a control site (e.g. in the case of transplanting lichens that can not be cultivated) are brought into the study site. A laboratory approach under fully controlled conditions for plant development and exposure to pollutants is also possible. This approach is mainly used to observe short-term impacts under specific exposure conditions.
The main limitation of biomonitoring lies in the biological nature of the bioindicator: success depends upon the sufficiently high presence of the organism studied, its sensitivity to other factors (biotic or abiotic) or even its competition with other organisms or species (Cuny, 2012). The effect of ecological factors can therefore be enough to result in a poor estimation of the effects of the pollutants studied.
Biomonitoring encompasses four concepts.
Bioindication occurs at the individual level and provides information on environmental impacts. It is based on the observation of the clinical and visible effects of pollution on an organism, part of an organism or a community of organisms (biocenosis). These effects are observable at the morphological, tissue or physiological level and include, for example, the appearance of leaf necrosis (Silva et al., 2012) or changes in root growth (Manier et al., 2009).
Necrosis caused by ozone on tobacco leaves (Photo courtesy of APPA).
Biointegration makes it possible to assess the long-term effects on communities by studying the density and specific diversity of populations within ecosystems. The sentinel species used are generally higher plants, bryophytes and lichens (Thimonier et al., 1994; Takahashi & Miyajima, 2010).
Bioaccumulation is the accumulation of contaminants in the tissues of plants or fungi, which then serve as a matrix to measure the levels of the different pollutants. This phenomenon of accumulation on the surface and/or inside the organism involves parameters of the external environment (e.g. environmental availability of pollutants) as well as physiological and metabolic components of the organism (e.g. translocation). Here, the tissue concentrations reflect the pollution levels of the environment and correspond to the bioavailable fraction which ultimately remains in the organism. These quantities are not therefore directly representative of those found in the surrounding environment. This method is widely used to study the impregnation of the environment by pollutants and to monitor their spatial and temporal evolution (Larsen et al., 2007; Cloquet et al., 2009; Gerdol et al., 2014).
The use of biomarkers facilitates the characterization of any
“Observable and/or measurable change at the molecular, biochemical, cellular or physiological level which reveals the current or previous exposure of an individual to at least one chemical substance of a polluting nature” (Lagadic et al., 1997).
A biomarker characterizes an early, invisible and specific stress effect at the infra-individual level. This is the most recent concept in the field of plant biomonitoring, and it is still being significantly developed. Biomarkers are used in domains including the monitoring of fluorescence in chlorophyll, photosynthetic activity (Catalyud & Barreno, 2004; Crous et al., 2006), enzyme activities (Rai & Agrawal, 2008), membrane integrity or genotoxicity (Rzepka & Cuny, 2011; Misik et al., 2011).
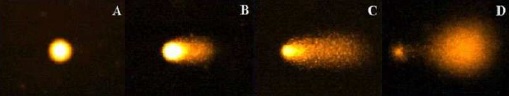
The comet test is used to evaluate the genotoxicity of a substance or an environment by measuring DNA damage (breakage). A: DNA intact, B and C: DNA damaged, D: DNA severely damaged (Gichner, 2003).
Biomonitoring with plants and fungi in the Nord-Pas-de-Calais
Air quality biomonitoring with plants and fungi
The main plant and fungi models used for air quality biomonitoring (Photo courtesy of APPA).
Lichens
Lichen biomonitoring has been used for many years by the LSVF, in collaboration with the APPA, to characterize and monitor the evolution of air quality in space and time (Delzenne-Van Haluwyn, 1973; Hardouin, 1983; Van Haluwyn, 1994; Van Haluwyn & Cottard, 1995; Cuny et al., 2004; Cuny et al., 2010).
Among the organisms used for air quality biomonitoring, epiphytic lichens are the most commonly used fungi. They are formed by the symbiotic association between a fungus and either a green alga (in 90% of cases) or a cyanobacterium (in 10% of cases). Their vegetative apparatus is a thallus (devoid of root system, stems or leaves) which also supports vegetative organs such as isidia and soralia, and organs for sexual reproduction (i.e. apothecia and perithecia).
Lichens develop slowly, with a growth that varies between less than one millimeter and one centimeter per year depending on the species and the living environment. They are entirely dependent on the atmosphere for their nutrition (Loppi & Nascimbene, 2010). In fact, most of the nutrients are taken from atmospheric deposition on the surface of the thallus (upper cortex: Nieboer et al., 1978). Unlike higher plants, they do not have a waxy cuticle or a stomata to modulate their exposure to atmospheric pollutants.
They are therefore very sensitive to substances that are present in the ambient air. Pollutants are thus easily absorbed by lichens and deeply disturb their metabolism (Conti & Cecchetti, 2001), sometimes resulting in visible morphological effects. The effects of air pollution can even modify the composition of lichenic communities according to the differential sensitivity of the species. Lichens are therefore ideal organisms for the observation when evaluating the effects of air quality on living organisms in time and space (Loppi et al., 2004).
Lichenic diversity, an ideal indicator of air quality (Photo courtesy of APPA).
Lichens are also excellent bioaccumulators due to their longevity, their annual biological activity and their fungal wall, which offering a multitude of possible sites for fixing pollutants (Loppi et al., 1997). This particularity makes it possible to measure the biological concentrations of bioaccumulative pollutants such as metallic trace elements, radioelements and persistent organic pollutants. Lichens are therefore excellent indicators of background air pollution when used as bio-integrators and bioaccumulators (Gadsdon et al., 2010).
The lichen Xanthoria parietina is widely used to evaluate bioaccumulation of persistent pollutants (Photo courtesy of APPA).
Regulation and standardization of air quality biomonitoring with plants and fungi
Directive 2004/107 / EC of the European Parliament concerning arsenic, cadmium, mercury, nickel and polycyclic aromatic hydrocarbons in ambient air is the only regulatory text that explicitly refers to biological methods for measurement of air quality. We can therefore consider that alongside physico-chemical studies, “the use of bio-indicators can be envisaged where regional models of the impact on ecosystems must be evaluated”.
Air quality biomonitoring methodology is based on the following standards, which are applicable at French and European levels:
- NF X43-900 Mai 2008 : Biosurveillance de l’air – Bio-indication de l’ozone par le tabac
- PR NF EN 16789 Novembre 2014 : Air ambiant – Biosurveillance à l’aide de plantes supérieures – Méthode de l’exposition normalisée du tabac (en projet : annulera et remplacera la norme NF X43-900:2008) ;
- NF X43-901 Mai 2008 : Biosurveillance de l’air – Biosurveillance active de la qualité de l’air à l’aide de ray-grass : des cultures à la préparation des échantillons ;
- NF X43-902 Mai 2008 : Biosurveillance de l’air – Biosurveillance passive de la qualité de l’air à l’aide de mousses autochtones : de la récolte à la préparation des échantillons
- NF X43-903 Mai 2008 : Biosurveillance de l’air – Détermination d’un indice biologique de lichens épiphytes (IBLE) ;
- NF X43-904 Janvier 2013 : Biosurveillance de l’air – Biosurveillance passive de la qualité de l’air à l’aide des lichens autochtones : de la récolte à la préparation des échantillons ;
- NF X43-905 Octobre 2014 : Biosurveillance passive de la pollution atmosphérique à partir de l’étude des dépôts particulaires foliaires ;
- NF EN 16413 Mars 2014 : Air Ambiant – Biosurveillance à l’aide de lichens – Évaluation de la diversité des lichens épiphytes – Qualité de l’air ;
- NF EN 16414 Mars 2014 : Air ambiant – Biosurveillance à l’aide de mousses – Accumulation des contaminants atmosphériques dans les mousses prélevées in situ : de la récolte à la préparation des échantillons ;
- XP X43-906 Août 2015 : Biosurveillance de l’air – Biosurveillance active de la qualité de l’air à l’aide de mousses ;
- en cours d’élaboration : PR NF X43-905 – Biosurveillance passive à partir de l’étude des dépôts particulaires foliaires.
References
Catalyud A & Barreno E. 2004. Response to ozone in two lettuce varieties on chlorophyll a fluorescence, photostnthetic pigments and lipids peroxidation. Plant Physiology and Biochemistry, 42: 549-555.
Cloquet C, De Muynck D, Signoret J, Vanhaecke F. 2009. Urban/peri-urban aerosol survey by determination of the concentration and isotopic composition of Pb collected by transplanted lichen Hypogymnia physodes. Environmental Science and Technology, 43 (3): 623-9.
Conti ME, Cecchetti G. 2001. Biological monitoring: lichens as bioindicators of air pollution assessment: a review. Environ Pollut, 114: 471-92.
Crous KI, Vandermeiren K, Ceulemans R. 2006. Physiological responses to cumulative ozone uptake in two white clover (Trifolium repens L. cv. Regal) clones with different ozone sensitivity. Environmental and Experimental Botany, 58(1-3): 169-179.
Cuny D, Davranche L, Thomas P, Kempa M, Van Haluwyn C. 2004. Spatial and temporal variations of trace element contents in Xanthoria parietina thalli collected in a highly industrialized area in Northern France as an element for a future epidemiological study. Journal of Atmospheric Chemistry, 49: 391-401.
Cuny D, Rzepka MA, Van Haluwyn C. 2008. Introduction à la biosurveillance végétale et fongique de la qualité de l’air, concept et intérêt. Spectra Analyse, 264: 31-35.
Cuny D., Quarre S., Devred I. et al. (2010). Évolution spatiale et temporelle des concentrations en Éléments Traces Métalliques de thalles de Xanthoria parietina récoltés sur la zone industrialo-portuaire de Dunkerque. Air Pur, n° 77, p. 43-49.
Cuny D. 2012. La biosurveillance végétale et fongique de la pollution atmosphérique : concepts et applications. Annales Pharmaceutiques Françaises, 70: 182—187.
Delzenne-Van Haluwyn C. 1973. Contribution à l’étude de la distribution des lichens épiphytes dans le Nord de la France : application au problème de la pollution atmosphérique. Thèse de doctorat, Faculté de Pharmacie, Lille: 162 p.
Gadsdon S, Dagley JR, Wolseley PA, Power SA. 2010. Relationships between lichen community composition and concentrations of NO2 and NH3. Environmental Pollution, 158: 2553-2560.
Garrec J-P, Van Haluwyn C. 2002. Biosurveillance végétale de la qualité de l’air. Tec & Doc, Paris.
Gerdol R, Marchesini R, Iacuminb P, Brancaleoni L. 2014. Monitoring temporal trends of air pollution in an urban area using mosses and lichens as biomonitors. Chemosphere, 108: 388-395.
Gichner T. 2003. DNA damage induced by indirect and direct acting mutagens in catalase-deficient transgenic tobacco Cellular and acellular Comet assays. Mutation Research 535: 187–193.
Hardouin M. (1983) Les lichens épiphytes de la région Sud-Est de Lille: généralités, distribution et estimation de la pollution atmosphérique acide. Thèse de Doctorat en Pharmacie. 137p.
Lagadic L, Caquet T, Amiard JC. 1997. Biomarqueurs en écotoxicologie : principes et définitions. Dans Lagadic L, Caquet, Amiard JC, Ramade F. Biomarqueurs en écotoxicologie – Aspects fondamentaux. Masson, Paris: 1-9.
Larsen RS, Bell JNB, James PW, Chimonides PJ, Rumsey FJ, Tremper A, Purvis OW. 2007. Lichen and bryophyte distribution on oak in London in relation to air pollution and bark acidity. Environmental Pollution, 146: 332-340.
Leblond S, Gombert-Courvoisier S, Louis-Rose S. 2014. Normalisation dans le domaine de la biosurveillance de la qualité de l’air. Workshop International « Biosurveillance végétale et fongique de la Qualité de l’Air ». Lille, France.
Loppi S, Nelli L, Ancora S, Bargagli R. 1997. Accumulation of trace elements in the peripheral and central parts of a foliose lichen thallus. Bryologist, 100: 251 –253.
Loppi S, Frati L, Paoli L, Bibabli V, Rossetti C, Bruscoli C, Corsini A. 2004. Biodiversity of epiphytic lichens and heavy metal contents of Flavoparmelia caperata thalli as indicators of temporal variations of air pollution in the town of Montecatini Terme (central Italy). Science of the Total Environment, 326: 113–122.
Loppi S. & Nascimbene J. (2010) Monitoring H2S air pollution caused by the industrial exploitation of geothermal energy: The pitfall of using lichens as bioindicators. Environmental Pollution 158 : 2635-2639.
Manier N, Deram A, Lecurieux F, Marzin D. 2009. Comparison between new wild plant Trifolium repens and Vicia faba on their sensitivity in detecting the genotoxic potential of heavy metal solutions and heavy metal-contaminated soils. Water, Air, and Soil Pollution, 202 (1-4): 343-352.
Misík M, Ma TH, Nersesyan A, Monarca S, Kim JK, Knasmueller S. 2011. Micronucleus assays with Tradescantia pollen tetrads: an update. Mutagenesis, 26(1): 215-21.
Niober EA, Richardson DHS, Tomassini FD. 1978. Mineral Uptake and Release by Lichens: An Overviewed. Bryologist, 81: 226-246.
Nylander W. 1866. Les lichens du Jardin du Luxembourg. Bulletin de la Société botanique de France, 13: 364‑372.
Rai R & Agrawal M. 2008. Evaluation of physiological and biochemical responses of two rice (Oryza sativa L.) cultivars to ambient air pollution using open top chambers at a rural site in India. Science of the Total Environment, 407 (1): 679-691.
Rzepka MA & Cuny D. 2011. Recherches préliminaires sur la mise en place dʼune biosurveillance végétale de la pollution atmosphérique en zone industrielle complexe : applications du ray-grass dans la zone industrialoportuaire de Dunkerque. Acta Botanica Gallica, 158(3): 387-400.
Silva DT, Meirelles ST, Moraes RM. 2012. Relationship between ozone, meteorological conditions, gas exchange and leaf injury in Nicotiana tabacum Bel-W3 in a sub-tropical region. Atmospheric Environment, 60: 211-216.
Takahashi T & Miyajima Y. 2010. Effects of roads on alpine and subalpine plant species distribution along an altitudinal gradient on Mount Norikura, central, Japan. Journal of Plant Research, 123: 741-749.
Thimonier A, Dupouey JL, Bost F, Becker M. 1994. Simultaneous eutrophication and acidification of a forest ecosystem in North-East France. New Phytologist, 126: 533-9.
Treshow, M. & Bell, J. N. B. 2002. Air pollution and plant life. Ed. John Wiley & Sons. Chichester, England. 467 pages. 2nd Ed.
Van Haluwyn C. (1994) Niveaux de la qualité de l’air sur le littoral Calais-Dunkerque ; évaluation en fonction de la diversité lichénique, Rapport SPPPI du Littoral. 57-74.
Van Haluwyn C. & Cottard S. (1995) Cartographie de la qualité de l’air de la communauté urbaine de Lille à partir de l’observation des lichens, Rapport AREMA LM. 14p.
Van Haluwyn C. 1998. La bioindication : applications dans notre région. Air Pur, 55: 23-25.
Van Haluwyn C, Cuny D, Garrec J-P. 2011. Introduction générale – Définitions, concepts et enjeux généraux de la biosurveillance de la qualité de lʼair. Pollution Atmosphérique, Numéro spécial : 7-13.